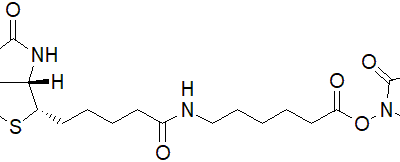Additional information
| Description | Sulfo-NHS-LC-biotin (Sulfosuccinimidyl 6-(biotinamido) Hexanoate) is an amine-reactive biotinylation agent known for its water solubility and polarity due to a sulfonate group on the NHS ring structure. This characteristic allows it to be directly used in aqueous reactions without prior dissolution in organic solvents. Upon reaction, Sulfo-NHS-LC-biotin forms a covalent amide linkage with amine groups in proteins and other molecules, releasing N-Hydroxysuccinimide. With a longer spacer length (22.4 Å) compared to NHS-biotin (13.5 Å), Sulfo-NHS-LC-biotin extends the reach of biotin probes, enhancing their ability to access deeply buried binding sites on protein surfaces. |
|---|---|
| Reference | [1] Bioconjugate Techniques , 2nd ed. By Greg T.Hermanson (Pierce Biotechnology, Thermo Fisher Scientific, Rockford, IL). Academic Press (an imprint of Elsevier): London, Amsterdam, Burlington, San Diego . 2008. ISBN 978-0-12-370501-3. |



![N-(3-(2-(2-(3-Aminopropoxy)ethoxy)ethoxy)propyl)-5-((3aS,4S,6aR)-2-oxohexahydro-1H-thieno[3,4-d]imidazol-4-yl)pentanamide](https://crolaboratories.com/wp-content/uploads/2022/09/B-22010-400x162.png)